In 2021, the CBSE introduced a new exam pattern for Class 10 Science due to the COVID-19 pandemic. The academic session was divided into Term 1 and Term 2, with separate sample papers and board exams for each term.

Term 1 featured only objective-type questions (MCQs), while Term 2 included short and long answer questions. This format aimed to reduce pressure on students and ensure fair assessment during uncertain times. On this page, you’ll find the complete CBSE Class 10 Science Sample Paper 2021 for both terms, along with detailed, easy-to-understand solutions in plain text format—perfect for board exam preparation.
CBSE Class 10 Science Sample Paper 2021 (Term 1 & Term 2) – Questions and Answers
Sample Question Paper (TERM – I) 2021-22
Class X
Science (086)
Time: 90 Minutes
- General Instructions: There is no negative marking.
- The Question Paper contains three sections.
- Section A has 24 questions. Attempt any 20 questions.
- Section B has 24 questions. Attempt any 20 questions.
- Section C has 12 questions. Attempt any 10 questions.
- All questions carry equal marks.
SECTION – A
Section – A consists of 24 questions. Attempt any 20 questions from this section.
The first attempted 20 questions would be evaluated.
Q1. Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of
Potassium Iodide solution to it. What would she observe?
A. The solution turned red.
B. Yellow precipitate was formed.
C. White precipitate was formed.
D. The reaction mixture became hot.
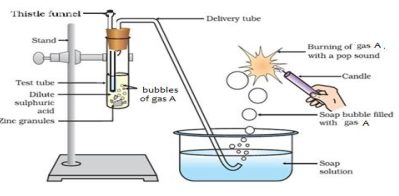
Q2. Identify gas A in the following experiment.
A. Nitrogen
B. Hydrogen
C. Oxygen
D. Carbon dioxide
Q3.
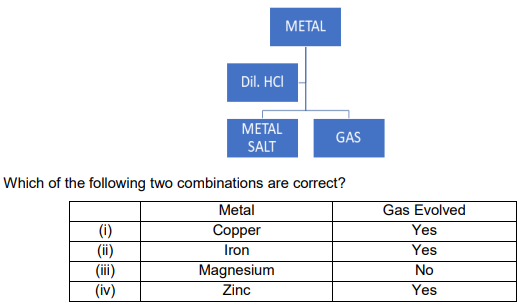
A. i and iii
B. i and iv
C. ii and iii
D. ii and iv
Q4. Which of the following correctly represents a balanced chemical equation?
A. Fe(s) + 4H2O(g) → Fe3O4 (s) + 4H2(g)
B. 3Fe(s) + 4H2O(g) → Fe3O4 (s) + 4H2(g)
C. 3Fe(s) + H2O(g) → Fe3O4 (s) + H2(g)
D. 3Fe(s) + 4H2O(g) → Fe3O4 (s) + H2(g)
5. The graph given below depicts a neutralization reaction (acid + alkali → salt + water).
The pH of a solution changes as we add excess of acid to an alkali.
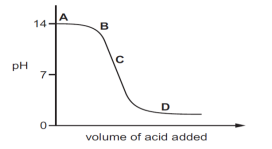
Which letter denotes the area of the graph where both acid and salt are present?
A. A
B. B
C. C
D. D
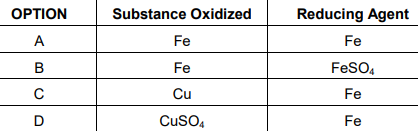
6. In the reaction of iron with copper sulphate solution:
CuSO4 + Fe —> Cu + FeSO4
Which option in the given table correctly represents the substance oxidised and the
reducing agent?
Q7. The chemical reaction between copper and oxygen can be categorized as:
A. Displacement reaction
B. Decomposition reaction
C. Combination reaction
D. Double displacement reaction
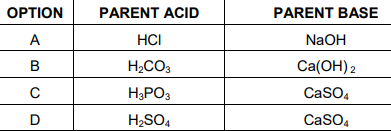
Q8. Which of the given options correctly represents the Parent acid and base of Calcium
Carbonate?
9. How will you protect yourself from the heat generated while diluting a concentrated acid?
A. By adding acid to water with constant stirring.
B. By adding water to acid with constant stirring.
C. By adding water to acid followed by base.
D. By adding base to acid with constant stirring.
10. Why is it important to balance a skeletal chemical equation?
A. To verify law of conservation of energy.
B. To verify the law of constant proportion.
C. To verify the law of conservation of mass.
D. To verify the l0aw of conservation of momentum.
Q11.Carefully study the diagram of the human respiratory system with labels A, B, C and D.
Select the option which gives correct identification and main function and /or
characteristic.
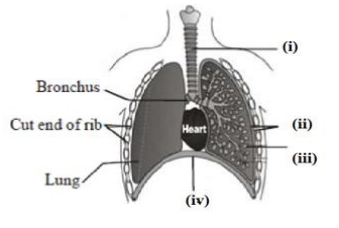
A. (i) Trachea: It is supported by bony rings for conducting inspired air.
B. (ii) Ribs: When we breathe out, ribs are lifted.
C. (iii) Alveoli: Thin-walled sac like structures for exchange of gases.
D. (iv) Diaphragm: It is pulled up when we breathe in.
Q12. Identify the option that indicates the correct enzyme that is secreted in location A, B and C.
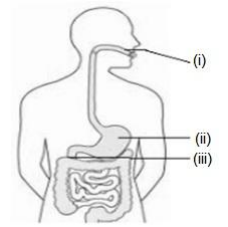
A. (i)-lipase, (ii)-trypsin, (iii)-pepsin
B. (i)-amylase, (ii)-pepsin, (iii)-trypsin
C. (i)-trypsin, (ii)-amylase, (iii)-carboxylase
D. (i)-permease, (ii)-carboxylase, (iii)-oxidase
Q13. Opening and closing of stomatal pore depends on:
A. Atmospheric temperature
B. oxygen concentration around stomata
C. carbon dioxide concentration around stomata
D. water content in the guard cells
14. The figure given below shows a schematic plan of blood circulation in humans with labels
(i) to (iv). Identify the correct label with its functions?
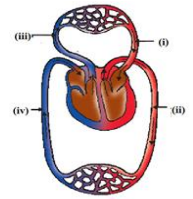
A. (i) Pulmonary vein – takes impure blood from body part.
B. (ii) Pulmonary artery – takes blood from lung to heart.
C. (iii) Aorta – takes blood from heart to body parts.
D. (iv) Vena cava takes – blood from body parts to right auricle.
Q15. Identify the phase of circulation which is represented in the diagram of heart given below.
Arrows indicate contraction of the chambers shown.

A. Blood transferred to the right ventricle and left ventricle simultaneously.
B. Blood is transferred to lungs for oxygenation and is pumped into various organs
simultaneously.
C. Blood transferred to the right auricle and left auricle simultaneously.
D. Blood is received from lungs after oxygenation and is received from various organs
of the body.
Q16. Observe the diagram of Human digestive system.
Match the labeling referred in column I and correlate with the function in column II.
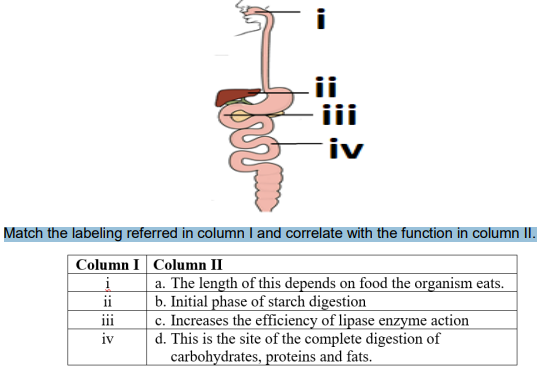
A. i.- a) ; ii – b) ; iii – c) ; iv- d)
B. i.- b) ; ii – c) ; iii – d) ; iv- a)
C. i.- b) ; ii – d) ; iii – c) ; iv- a)
D. i.- d) ; ii – a) ; iii – b) ; iv- c)
17. Which of the following mirror is used by a dentist to examine a small cavity in a patient’s
teeth?
A. Convex mirror
B. Plane mirror
C. Concave mirror
D. Any spherical mirror
Q18.
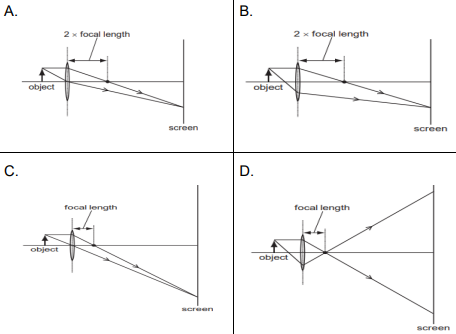
Q19. Which of the following can make a parallel beam of light when light from a point source is
incident on it?
A. Concave mirror as well as convex lens.
B. Convex mirror as well as concave lens.
C. Two plane mirrors placed at 90° to each others.
D. Concave mirror as well as concave lens.
20. Consider these indices of refraction: glass: 1.52; air: 1.0003; water: 1.333. Based on the
refractive indices of three materials, arrange the speed of light through them in
decreasing order.
A. The speed of light in water > the speed of light in air > the speed of light in glass.
B. The speed of light in glass > the speed of light in water > the speed of light in air.
C. The speed of light in air > the speed of light in water > the speed of light in glass.
D. The speed of light in glass > the speed of light in air > the speed of light in water.
21. If a beam of red light and a beam of violet light are incident at the same angle on the
inclined surface of a prism from air medium and produce angles of refraction r and v
respectively, which of the following is correct?
A. r = v
B. r > v
C. r = 1/v
D. r < v
22.
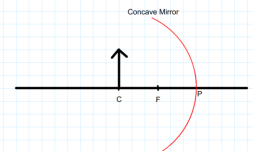
Examine the above figure and state which of the following option is correct?
[one small box in the figure is equal to 1 cm]
A. The mirror has a focal length of -6 cm and will produce an image of magnification +1.
B. The mirror has a focal length of -3 cm and will produce an image of magnification -1.
C. The mirror has a focal length of -3 cm and will produce an image of magnification +1.
D. The mirror has a focal length of -6 cm and will produce an image of magnification -1
23.
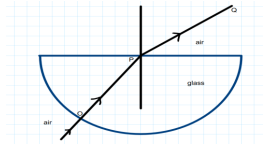
The angle of incidence from air to glass at the point O on the hemispherical glass slab is.
A. 45°
B. 0°
C. 90°
D. 180°
24 A prism ABC (with BC as base) is placed in different orientations. A narrow beam of
white light is incident on the prism as shown in below Figure. In which of the following
diagrams, after dispersion, the third colour from the top of the spectrum corresponds to
the colour of the sky?
A. (i)
B. (ii)
C. (iii)
D. (iv)
SECTION – B
Section – B consists of 24 questions (Sl. No.25 to 48). Attempt any 20 questions from this section.
The first attempted 20 questions would be evaluated.
25.
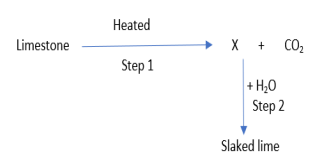
Identify the correct option from the given table which represents the type of reactions
occurring in step 1 and step 2.
26. In which year is concentration of hydrogen ion the highest?
A. 2002
B. 2008
C. 2011
D. 2005
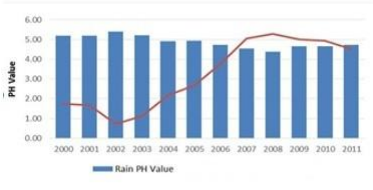
27. The diagram shows the reaction between metal and dil. acid.
What is the reason for different behaviour of Mg in test tube B?
A. Mg is lighter element than dil. HCl
B. Mg reacts with dil. HCl to produce H2 gas which helps in floating
C. Mg reacts with dil. HCl to produce N2 gas which helps in floating
D. Mg reacts with dil. HCl to produce CO2 gas which helps in floating
28. The table shown below gives information about four substances: A, B, C and D.
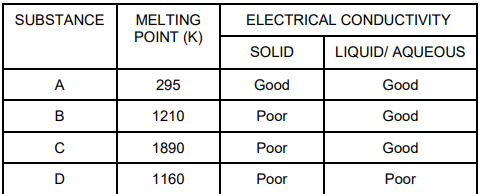
Identify Ionic compounds from the above given substances.
A. A, B
B. B, C
C. A, B, D
D. A, C, D
29. Vinay observed that the stain of curry on a white shirt becomes reddish-brown when
soap is scrubbed on it, but it turns yellow again when the shirt is washed with plenty of
water. What might be the reason for his observation?
i. Soap is acidic in nature
ii. Soap is basic in nature
iii. Turmeric is a natural indicator which gives reddish tinge in bases
iv. Turmeric is a natural indicator which gives reddish tinge in acids
A. i and ii
B. ii and iii
C. i and iv
D. ii and iv
30. In which of the following setups would the bulb glow?
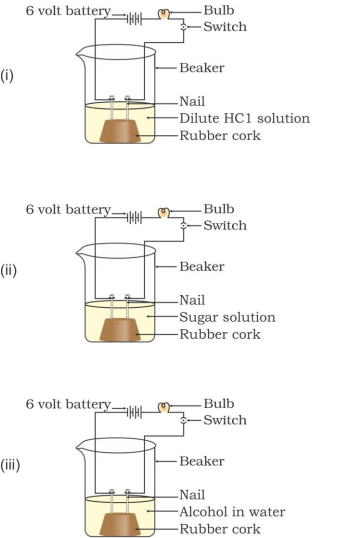
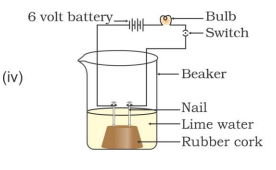
A
. i and ii
B
. i and iv
C
. ii, iii and iv
D
. i, ii and iv
Question No. 31 to 35 consist of two statements – Assertion (A) and Reason (R). Answer these
questions selecting the appropriate option given below:
A. Both A and R are true and R is the correct explanation of A
B. Both A and R are true and R is not the correct explanation of A
C. A is true but R is false
D. A is False but R is true
- Assertion: Fresh milk in which baking soda is added, takes a longer time to set as curd.
Reason: Baking soda decreases the pH value of fresh milk to below 6. - Assertion: Decomposition of vegetable matter into compost is an endothermic reaction.
Reason: Decomposition reaction involves breakdown of a single reactant into simpler
products. - Assertion: Resins and gums are stored in old xylem tissue in plants.
Reason:Resins and gums facilitate transport of water molecules. - Assertion: Sky appears blue in the day time.
Reason: White light is composed of seven colours. - The table given below shows the reaction of a few elements with acids and bases to H2
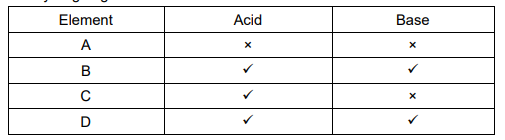
Which of these elements form amphoteric oxides?
A. A and D
B. B and D
C. A and C
D. B and D
36.
In which of the following groups of organisms, blood flows through the heart only once
during one cycle of passage through the body?
A. Rabbit, Parrot, Turtle
B. Frog, crocodile, Pigeon
C. Whale, Labeo, Penguin
D. Shark, dog fish, sting ray
- What is common between extensive network of blood vessels around walls of alveoli and
in glomerulus of nephron?
A. Thick walled arteries richly supplied with blood
B. Thin walled veins poorly supplied with blood
C. Thick walled capillaries poorly supplied with blood.
D. Thin walled capillaries richly supplied with blood - Plants use completely different process for excretion as compared to animals. Which one
of the following processes is NOT followed by plants for excretion?
A. They can get rid of excess water by transpiration.
B. They selectively filter toxic substances through their leaves.
C. Waste products are stored as resins and gums in old xylem.
D. They excrete waste substances into the soil around them. - If the power of a lens is – 4.0 D, then it means that the lens is a
A. concave lens of focal length -50 m
B. convex lens of focal length +50 cm
C. concave lens of focal length -25 cm
D. convex lens of focal length -25 m - Rays from Sun converge at a point 15 cm in front of a concave mirror. Where should an
object be placed so that size of its image is equal to the size of the object?
A. 30 cm in front of the mirror
B. 15 cm in front of the mirror
C. Between 15 cm and 30 cm in front of the mirror
D. More than 30 cm in front of the mirror
- In which of the following groups of organisms, food material is broken down outside the
body and then absorbed in?
A. mushroom, green plants, amoeba
B. yeast, mushroom, bread mould
C. paramecium, amoeba, cuscuta
D. cuscuta, lice, tapeworm - In a person the tubule part of the nephron is not functioning at all. What will its effect be
on urine formation?
A. The urine will not be formed.
B. Quality and quantity of urine is unaffected.
C. Urine is more concentrated.
D. Urine is more diluted. - If the real image of a candle flame formed by a lens is three times the size of the flame
and the distance between lens and image is 80 cm, at what distance should the candle
be placed from the lens?
A. -80cm
B. -40 cm
C. -40/3 cm
D. -80/3 cm
44. While looking at the above diagram, Nalini concluded the following-
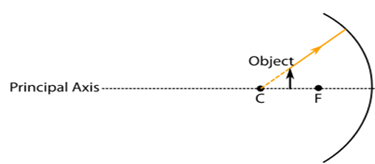
i. the image of the object will be a virtual one.
ii. the reflected ray will travel along the same path as the incident ray but in opposite
direction.
iii. the image of the object will be inverted.
iv. this is a concave mirror and hence the focal length will be negative.
Which one of the above statements are correct?
A. i and ii
B. i and iii
C. ii, iii and iv
D. i, ii, iii and iv
45.
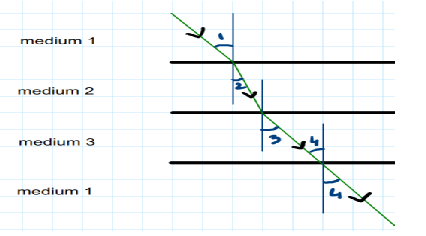
In the above diagram light is travelling through different media. It is noted by a scientist
that ∠1= ∠3= ∠4 but ∠2 <∠1. Which of the following statement would be correct?
A. Medium 1 is the denser than medium 3 but it’s density is equal to medium 2.
B. Medium 2 is the rarest medium.
C. Medium 3 is denser than medium 1.
D. Medium 1 and 3 are essentially the same medium, but medium 2 is denser than 1
and 3.
46. The refractive index of flint glass is 1.65 and that for alcohol is 1.36 with respect to air.
What is the refractive index of the flint glass with respect to alcohol ?
A. 0.82
B. 1.21
C. 1.11
D. 1.01
47.
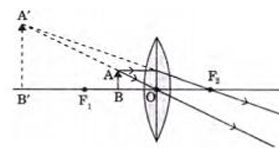
The above lens has a focal length of 10 cm. The object of height 2 mm is placed at a
distance of 5 cm from the pole. Find the height of the image.
A. 4 cm
B. 6.67 mm
C. 4 mm
D. 3.33 mm
48. A cable manufacturing unit tested few elements on the basis of their physical properties.
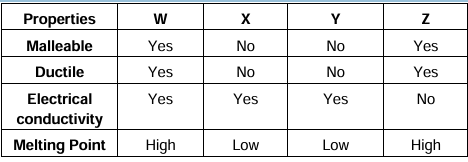
Which of the above elements were dicarded for usage by the company?
A. W, X, Y
B. X, Y, Z
C. W, X, Z
D. W, X, Z
SECTION – C
Section- C consists of three Cases followed by questions. There are a total of 12 questions in this
section. Attempt any 10 questions from this section.
The first attempted 10 questions would be evaluated.
Case The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third
largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious
process and the salt obtained is used as raw materials for manufacture of various sodium
compounds.
One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and
in soda acid fire extinguishers.
The table shows the mass of various compounds obtained when 1litre of sea water is
evaporated
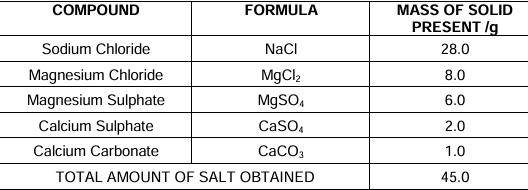
- Which compound in the table reacts with acids to release carbon dioxide?
A. NaCl
B. CaSO4
C. CaCO3
D. MgSO4 - How many grams of Magnesium Sulphate are present in 135g of solid left by evaporation
of sea water?
A. 6g
B. 12g
C. 18g
D. 24g - What is the saturated solution of Sodium Chloride called?
A. Brine
B. Lime water
C. Slaked lime
D. Soda water - What is the pH of the acid which is used in the formation of common salt?
A. Between 1 to 3
B. Between 6 to 8
C. Between 8 to 10
D. Between 11 to 13
Case The Figure shown below represents an activity to prove the requirements for
photosynthesis. During this activity, two healthy potted plants were kept in the dark for 72
hours. After 72 hours, KOH is kept in the watch glass in setup X and not in setup Y. Both
these setups are air tight and have been kept in light for 6 hours. Then, Iodine Test is
performed with one leaf from each of the two plants X and Y. 18 - This experimental set up is used to prove essentiality of which of the following
requirements of photosynthesis?
A. Chlorophyll
B. Oxygen
C. Carbon dioxide
D. Sunlight - The function of KOH is to absorb
A. Oxygen.
B. Carbon dioxide.
C. Moisture.
D. Sunlight. - Which of the following statements shows the correct results of Iodine Test performed on
the leaf from plant X and Y respectively?
A. Blue – black colour would be obtained on the leaf of plant Xand no change in colour
on leaf of plant Y.
B. Blue – black colour would be obtained on the leaf of plant Y and no change in colour
onleaf of plant X.
C. Red colour would be obtained on the leaf of plant X and brown colour on the leaf of
plant Y.
D. Red colour would be obtained on the leaf of plant Y and brown colour on the leaf of
plant X. - Which of the following steps can be followed for making the apparatus air tight?
i. placing the plants on glass plate
ii. using a suction pump.
iii. applying aseline to seal the bottom of jar.
iv. creating vacuum
A. i and ii
B. ii. and iii
C. i. and iii
D. ii. and iv
Case Noor, a young student, was trying to demonstrate some properties of light in her Science
project work. She kept ‘X’ inside the box (as shown in the figure) and with the help of a
laser pointer made light rays pass through the holes on one side of the box. She had a
small butter-paper screen to see the spots of light being cast as they emerged
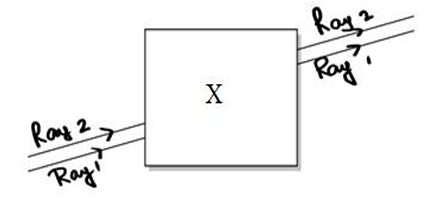
- What could be the ‘X’ that she placed inside the box to make the rays behave as shown?
A. a converging lens
B. a parallel-sided glass block
C. a plane mirror
D. a triangular prism - She measured the angles of incidence for both the rays on the left side of the box to be
48.60. She knew the refractive index of the material ‘X’ inside the box was 1.5. What will
be the approximate value of angle of refraction?
A. 450
B. 400
C. 300
D. 600
(use the value: sin 48.60 ≈ 0.75) - Her friend noted the following observations from this demonstration:
i. Glass is optically rarer than air.
ii. Air and glass allow light to pass through them with the same velocity.
iii. Air is optically rarer than glass.
iv. Speed of light through a denser medium is faster than that of a rarer medium.
v. The ratio: sin of angle of incidence in the first medium to the ratio of sin of angle of
refraction in the second medium, gives the refractive index of the second material
with respect to the first one.
Which one of the combination of the above statements given below is correct.
A. ii, iv and v are correct.
B. iii and iv are correct.
C. i, iv and v are correct.
D. iii and v are correct.
60. If the object inside the box was made of a material with a refractive index less than 1.5
then the
A. lateral shift of the rays would have been less.
B. lateral shift of the rays would have been more.
C. lateral shift of the rays would remain the same as before.
D. there is not enough information to comment on any of the above statements
TERM 2 – Subjective Questions and Answers
SAMPLE QUESTION PAPER
CLASS X
Science (086)
Term 2 (2021-22)
Max. Marks:40 Time allowed: 2 hours
General Instructions:
i) All questions are compulsory.
ii) The question paper has three sections and 15 questions. All questions are compulsory.
iii) Section–A has 7 questions of 2 marks each; Section–B has 6 questions of 3 marks each; and
Section–C has 2 case based questions of 4 marks each.
iv) Internal choices have been provided in some questions. A student has to attempt only one of
the alternatives in such questions.
SECTION – A
1 The table shows the electronic structures of four elements.
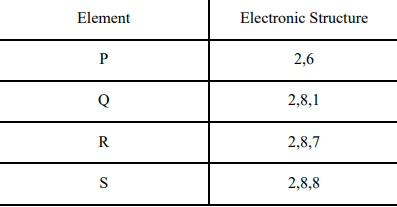
a. Identify which element(s) will form covalent bonds with carbon.
b. “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason. 2
The diagram below shows part of the periodic table.
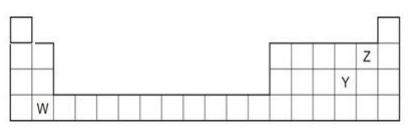
a. Which elements would react together to form covalent compounds?
b. Between the two elements W and Z, which will have a bigger atomic radius? Why?a. Identify which element(s) will form covalent bonds with carbon.
3.a. Trace the path a male gamete takes to fertilise a female gamete after being released from the penis.
b. State the number of sets of chromosomes present in a zygote.
4. Rajesh observed a patch of greenish black powdery mass on a stale piece of bread.
a. Name the organism responsible for this and its specific mode of asexual reproduction.
b. Name its vegetative and reproductive parts.
5. Mustard was growing in two fields- A and B. While Field A produced brown coloured seeds, field B produced yellow coloured seeds. It was observed that in field A, the offsprings showed only the parental trait for consecutive generations, whereas in field B, majority of the offsprings showed a variation in the progeny..What are the probable reasons for these?
OR
In an asexually reproducing species, if a trait X exists in 5% of a population and trait Y exists in 70% of the same population, which of the two trait is likely to have arisen earlier? Give reason.
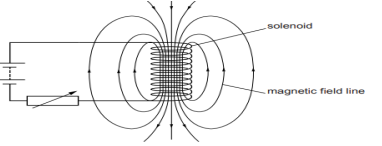
6. A circuit contains a battery, a variable resistor and a solenoid. The figure below shows the magnetic field pattern produced by the current in the solenoid.
a. State how the magnetic field pattern indicates regions where the magnetic field is stronger.
b. What happens to the magnetic field when the current in the circuit is reversed?
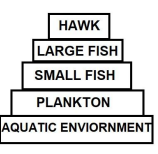
7 DDT was sprayed in a lake to regulate breeding of mosquitoes. How would it affect the trophic levels in the following food chain associated with a lake? Justify your answer.
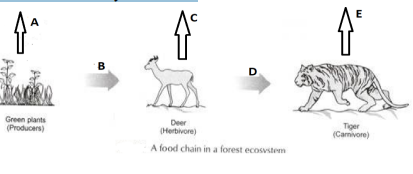
OR
In the following food chain, vertical arrows indicate the energy lost to the environment and horizontal arrows indicate energy transferred to the next trophic level. Which one of the three vertical arrows (A, C and E) and which
one of the two horizontal arrows (B and D) will represent more energy transfer? Give reason for your answer.
SECTION – B
8. Choose an element from period 3 of modern periodic table that matches the
description given below in each instance. Give reason for your choice.
a. It has a similar structure to diamond.
b. It has same valency as Lithium.
c. It has variable valency and is a member of the Oxygen family (group 16). 3
9. a. How many isomers are possible for the compound with the molecular formula C4H8?
Draw the electron dot structure of branched chain isomer.
b. How will you prove that C4H8 and C5H10 are homologues?
OR
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
a. Identify ‘A’ and draw its electron dot structure.
b. Give the molecular formulae of any two homologues of ‘A’.
10. Two pea plants – one with round yellow seeds (RRYY) and another with .wrinkled green (rryy) seeds produce F1 progeny that have round, yellow (RrYy) seeds.
When F1 plants are self-pollinated, which new combination of characters is expected in F2 progeny? How many seeds with these new combinations of characters will be produced when a total 160 seeds are produced in F2
generation? Explain with reason.
3
11 a. It would cost a man Rs. 3.50 to buy 1.0 kW h of electrical energy from the
Main Electricity Board. His generator has a maximum power of 2.0 kW.
The generator produces energy at this maximum power for 3 hours.
Calculate how much it would cost to buy the same amount of energy from
the Main Electricity Board.(1 Mark)
b. A student boils water in an electric kettle for 20 minutes. Using the same
mains supply he wants to reduce the boiling time of water. To do so should
he increase or decrease the length of the heating element? Justify your
answer.(2 Marks)
12.
13 Gas A, found in the upper layers of the atmosphere, is a deadly poison but is essential for all living beings. The amount of this gas started declining sharply in the 1980s.
a. Identify Gas A. How is it formed at higher levels of the atmosphere?
b. Why is it essential for all living beings? State the cause for the depletion of this gas.
SECTION – C
This section has 02 case-based questions (14 and 15). Each case is followed by 03 sub-questions
(a, b and c). Parts a and b are compulsory. However, an internal choice has been provided in part c.
14. Sahil performed an experiment to study the inheritance pattern of genes. He crossed tall pea plants (TT) with short pea plants (tt) and obtained all tall plants in F1 generation.
a. What will be set of genes present in the F1 generation? (1 Mark)
b. Give reason why only tall plants are observed in F1 progeny. (1 Mark)
c. When F1 plants were self – pollinated, a total of 800 plants were produced. How many of these would be tall, medium height or short plants? Give the genotype of F 2 generation. (2 Marks)
OR
When F1 plants were cross – pollinated with plants having tt genes, a total
of 800 plants were produced. How many of these would be tall, medium
height or short plants? Give the genotype of F 2 generation. 4
15.Ansari Sir was demonstrating an experiment in his class with the setup as
shown in the figure below.
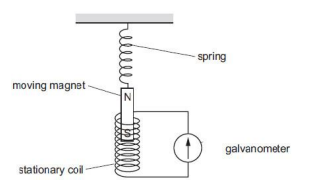
A magnet is attached to a spring. The magnet can go in and out of the stationary coil. He lifted the Magnet and released it to make it oscillate through the coil. Based on your understanding of the phenomenon, answer the following questions.
a. What is the principle which Ansari Sir is trying to demonstrate?
b. What will be observed when the Magnet starts oscillating through the coil. Explain the reason behind this observation.
c. Consider the situation where the Magnet goes in and out of the coil. State two changes which could be made to increase the deflection in the galvanometer.
OR
Is there any difference in the observations in the galvanometer when the
Magnet swings in and then out of the stationary coil? Justify your answer
For the official Class 10 Mathematics Solutions, you can visit:
- NCERT Textbooks (for Class 10):
Class-wise Solutions
Class 12:
Class 12 Physics – NCERT Solutions
Class 12 Chemistry – NCERT Solutions
Class 11:
- Class 11 Physics – NCERT Solutions
- Class 11 Chemistry – NCERT Solutions
- Class 11 Biology – NCERT Solutions
- Class 11 Math – NCERT Solutions
Class 10:
Class 9:
Class 8:
Class 7:
Class 6:
Subject-wise Solutions
Physics:
Chemistry:
Biology:
Math:
- Class 11 Math – NCERT Solutions
- Class 10 Math – NCERT Solutions
- Class 9 Math – NCERT Solutions
- Class 8 Math – NCERT Solutions
Science:
- Class 10 Science – NCERT Solutions
- Class 9 Science – NCERT Solutions
- Class 8 Science – Oxford Solutions
- Class 7 Science – Oxford Solutions
- Class 6 Science – Oxford Solutions
NEET BIOLOGY
- Evolution
- Breathing and Exchange of Gases
- Anatomy of Flowering Plants
- Body Fluids and Circulation
- Human Health and Disease
- Microbes in Human Welfare
- Cell Cycle and Cell Division
- Biotechnology and Its Applications
- Biodiversity and Conservation
- Morphology of Flowering Plants
Practicing with the CBSE Class 10 Science Sample Paper 2021 is one of the smartest ways to prepare for the board exam, especially considering the changed exam pattern due to the COVID-19 pandemic. The division into Term 1 and Term 2 gave students a different experience, and solving both sample papers helps build familiarity with this unique structure. Whether it’s mastering objective-type MCQs or writing accurate descriptive answers, these papers offer the right practice for every section of the syllabus.
The CBSE Class 10 Science Sample Paper 2021 includes well-crafted questions based on real-life applications, case studies, and experimental concepts from Physics, Chemistry, and Biology. With clearly explained answers provided for each question, students can evaluate their preparation and correct their mistakes effectively.
Teachers and tutors can also use the CBSE Class 10 Science Sample Paper 2021 as a revision tool in classrooms or online classes. It serves as a blueprint for understanding the marking scheme and question weightage, helping students write answers as expected by CBSE examiners.
In conclusion, the CBSE Class 10 Science Sample Paper 2021 is more than just a practice tool — it’s a complete guide for exam success. By solving both Term 1 and Term 2 papers with the correct approach, students can strengthen their concepts, improve answer-writing skills, and gain the confidence needed to score high marks in the final board exam.
