CBSE Class 10 Science Sample Paper 2020
CBSE Board – Free PDF download and interactive content.
High-Yield MCQs
Curated questions designed for quick revision and maximum scoring.
Detailed Solutions
Every answer includes clear explanation and concept recap.
Chapter-Wise Format
Structured exactly as per syllabus for focused practice.
Ready to Boost Your Score?
Download Now & Start PracticingSample Question Paper 2020-21
Class X
Science (086) Theory
Time: 3 Hours Maximum Marks: 80
General Instructions:
(i) The question paper comprises four sections A, B, C and D. There are 36 questions in the question paper. All questions are compulsory.
(ii) Section–A – question no. 1 to 20 – all questions and parts thereof are of one mark each. These questions contain multiple choice questions (MCQs), very short answer questions and assertion – reason type questions.
(iii) Section–B – question no. 21 to 26 are short answer type questions, carrying 2 marks each. Answers to these questions should in the range of 30 to 50 words.
(iv) Section–C – question no. 27 to 33 are short answer type questions, carrying 3 marks each. Answers to these questions should in the range of 50 to 80 words.
(v) Section–D – question no. – 34 to 36 are long answer type questions carrying 5 marks each. Answer to these questions should be in the range of 80 to 120 words.
(vi) There is no overall choice. However, internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
(vii) Wherever necessary, neat and properly labeled diagrams should be drawn
SECTION A
Q1. List any two observations when Ferrous Sulphate is heated in a dry test tube?
Answer:
- The green colour of ferrous sulphate crystals changes to brown.
- A pungent smell of sulphur dioxide and sulphur trioxide gases is observed.
Reason:
When ferrous sulphate (FeSO₄·7H₂O) is heated, it first loses water and then decomposes to form ferric oxide (Fe₂O₃), which is brown in colour, along with SO₂ and SO₃ gases.
OR
Identify the products formed when 1 mL of dilute Hydrochloric acid is added to 1 g of Sodium metal?
Answer:
The products formed are Sodium chloride (NaCl) and Hydrogen gas (H₂).
Chemical Equation:
Na + HCl → NaCl + H₂ ↑
Reason:
Sodium is a highly reactive metal. It reacts vigorously with dilute hydrochloric acid to form salt (sodium chloride) and releases hydrogen gas.
Q2. Write the chemical name and chemical formula of the salt used to remove permanent hardness of water.
Answer:
Chemical Name: Sodium carbonate (Washing soda)
Chemical Formula: Na₂CO₃·10H₂O
Reason:
Washing soda reacts with calcium and magnesium salts present in hard water and removes permanent hardness.
Q3. Which of the following is not observed in a homologous series? Give reason.
Answer:
Correct option: (a) Change in chemical properties
Reason:
Members of a homologous series have the same functional group, so they show similar chemical properties, not different ones.
Q4. Why does the Sun appear white at noon?
Answer:
At noon, the Sun is overhead and sunlight travels the shortest distance through the atmosphere.
Reason:
Very little scattering of light occurs, so all colours reach our eyes together, making the Sun appear white.
Q5. Both a spherical mirror and a thin spherical lens have a focal length of (-)15 cm. What type are they?
Answer:
- Mirror: Concave mirror
- Lens: Concave lens
Reason:
Negative focal length indicates a concave mirror and a concave (diverging) lens.
Q6. The image formed by a concave mirror is real, inverted and larger than the object. Where is the object placed?
Answer:
The object is placed between the focus (F) and the centre of curvature (C).
Reason:
In this position, a concave mirror forms a real, inverted, and magnified image.
OR
Name the part of a lens through which a ray passes without deviation.
Answer:
Optical centre
Reason:
A ray passing through the optical centre of a lens emerges without bending.
Q7. What are the two observations from the galvanometer reading?
Answer:
- The galvanometer shows a momentary deflection when the key is inserted.
- It shows a deflection in the opposite direction when the key is removed.
Reason:
This happens due to electromagnetic induction. A changing current in one coil induces current in the other coil only when the circuit is made or broken.
Q8. Draw the magnetic field lines around a straight current carrying conductor.
Answer
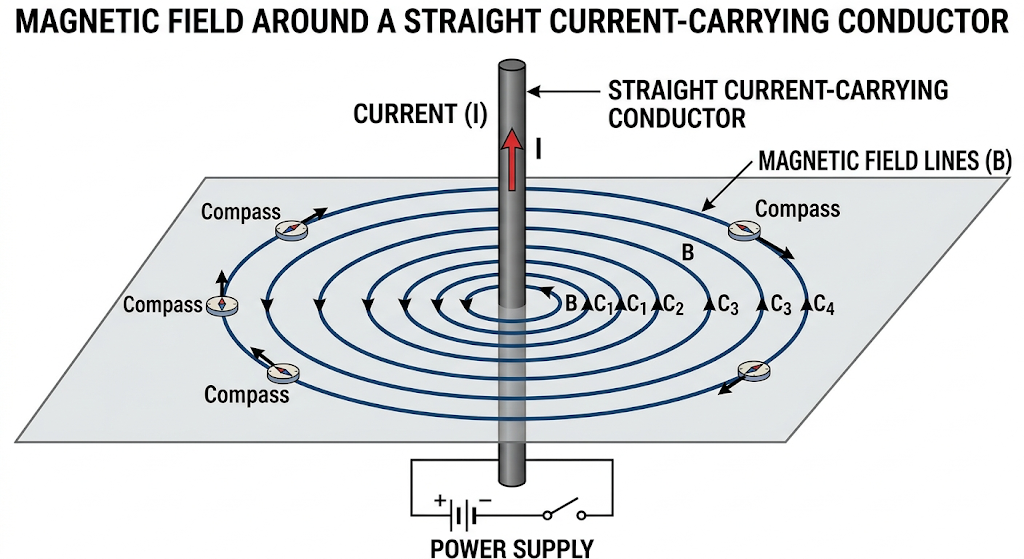
Magnetic field lines are concentric circles around the conductor.
Direction:
Given by the right-hand thumb rule (thumb → current, fingers → field direction).
Q9. Two unequal resistances are connected in parallel. What can be said about voltage drop?
Answer:
The voltage drop across both resistors is the same.
Reason:
In parallel combination, all resistors are connected across the same two points, so potential difference remains equal.
OR
Work done to move charge Q from infinity to point A (potential V)?
Answer:
W = QV
Reason:
Work done is equal to the product of charge and potential.
Q10. Veins are thin walled and have valves. Justify.
Answer:
Veins carry blood at low pressure, so they have thin walls.
They contain valves to prevent backflow of blood.
Q11. How is small intestine adapted for absorption?
Answer:
The inner lining has villi which increase surface area for absorption.
Reason:
More surface area allows efficient absorption of digested food.
OR
Which has longer small intestine: goat or tiger?
Answer:
Goat
Reason:
Goat is a herbivore and needs longer intestine to digest cellulose, while tiger (carnivore) has shorter intestine.
Q12. How ozone performs essential function despite being poisonous?
Answer:
Ozone forms a layer in the upper atmosphere.
Reason:
It absorbs harmful ultraviolet (UV) radiation from the Sun and protects living organisms.
OR
Why food chain cannot have more than four trophic levels?
Answer:
Because energy decreases at each trophic level.
Reason:
Only about 10% energy is transferred to the next level, so higher levels cannot be sustained.
Q13. State the role of pancreas in digestion.
Answer:
Pancreas secretes pancreatic juice.
Reason:
It contains enzymes like amylase, trypsin, and lipase which help in digestion of carbohydrates, proteins, and fats.
For question numbers 14, 15 and 16, two statements are given- one labeled Assertion (A) and the other labeled Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below:
a) Both A and R are true, and R is correct explanation of the assertion.
b) Both A and R are true, but R is not the correct explanation of the assertion.
c) A is true, but R is false.
d) A is false, but R is true.
14. Assertion: After white washing the walls, a shiny white finish on walls is obtained after two to three days.
Reason: Calcium Oxide reacts with Carbon dioxide to form Calcium Hydrogen Carbonate which gives shiny white finish.
15. Assertion: Food chain is responsible for the entry of harmful chemicals in our bodies.
Reason: The length and complexity of food chains vary greatly.
OR
Assertion: Greater number of individuals are present in lower trophic levels.
Reason: The flow of energy is unidirectional.
16. Assertion: A geneticist crossed a pea plant having violet flowers with a pea plant with white flowers, he got all violet flowers in first generation.
Reason: White colour gene is not passed on to next generation.
Answer:
Q14
Assertion (A): True
Reason (R): False
Correct Option: (c) A is true, but R is false.
Explanation:
After whitewashing, calcium hydroxide reacts with carbon dioxide to form calcium carbonate (CaCO₃), which gives the shiny white finish.
The reason is incorrect because it mentions calcium hydrogen carbonate, which is not responsible for the white finish.
Q15 (Option 1)
Assertion (A): True
Reason (R): False
Correct Option: (c) A is true, but R is false.
Explanation:
Food chains cause biomagnification, leading to entry of harmful chemicals into our bodies.
But the reason about length and complexity of food chains does not explain this.
OR (Q15 Alternative)
Assertion (A): True
Reason (R): True
Correct Option: (b) Both A and R are true, but R is not the correct explanation of A.
Explanation:
Lower trophic levels have more individuals due to higher energy availability.
Although energy flow is unidirectional, it does not directly explain the number of individuals.
Q16
Assertion (A): True
Reason (R): False
Correct Option: (c) A is true, but R is false.
Explanation:
Violet flower colour is dominant, so all F₁ generation plants appear violet.
The white gene is not absent—it is recessive and present but not expressed.
Answer Q. No 17 – 20 contain five sub-parts each. You are expected to answer any four subparts in these questions.
17 Read the following and answer any four questions from 17 (i) to 17 (v)
All living cells require energy for various activities. This energy is available by the breakdown of simple carbohydrates either using oxygen or without using oxygen.
(i) Energy in the case of higher plants and animals is obtained by
a) Breathing
b) Tissue respiration
c) Organ respiration
d) Digestion of food
(ii) The graph below represents the blood lactic acid concentration of an athlete during a race of 400 m and shows a peak at point D.
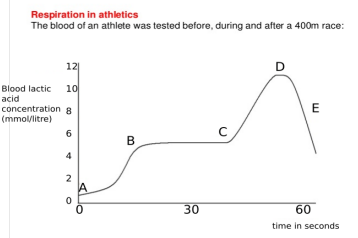
Lactic acid production has occurred in the athlete while running in the 400 m race. Which of the following processes explains this event?
a) Aerobic respiration
b) Anaerobic respiration
c) Fermentation
d) Breathing
(iii) Study the graph below that represents the amount of energy supplied with respect to the time while an athlete is running at full speed.
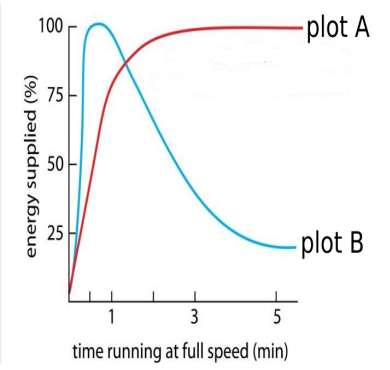
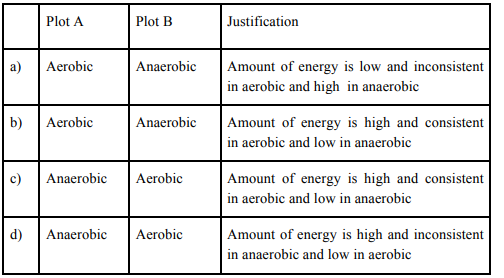
Choose the correct combination of plots and justification provided in the following table.
(iv) The characteristic processes observed in anaerobic respiration are
i) presence of oxygen
ii) release of carbon dioxide
iii) release of energy
iv) release of lactic acid
a) i) ,ii) only
b) i), ii), iii) only
c) ii), iii), iv) only
d) iv) only
(v) Study the table below and select the row that has the incorrect information
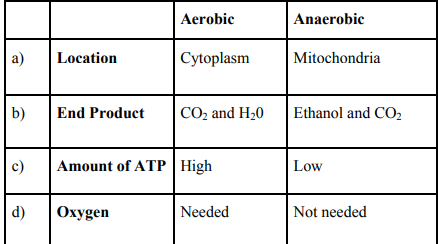
18. Read the following and answer any four questions from 18 (i) to 18 (v). Metallic Character
The ability of an atom to donate electrons and form positive ion (cation) is known as electropositivity or metallic character. Down the group, metallic character increases due to increase in atomic size and across the period, from left to right electropositivity decreases due to decrease in atomic size. Non-Metallic Character The ability of an atom to accept electrons to form a negative ion (anion) is called non-metallic character or electronegativity. The elements having high electro-negativity have a higher tendency to gain electrons and form anion. Down the group, electronegativity decreases due to increase in atomic size and across the period, from left to right electronegativity increases due to decrease in atomic size.
18 (i) Which of the following correctly represents the decreasing order of metallic character of Alkali metals plotted in the graph?
a) Cs>Rb>Li>Na>K
b) K>Rb>Li>Na>Cs
c) Cs>Rb>K>Na>Li
d) Cs>K>Rb>Na>Li
18 (ii) Hydrogen is placed along with Alkali metals in the modern periodic table though it shows non-metallic character
a) as Hydrogen has one electron & readily loses electron to form negative ion
b) as Hydrogen can easily lose one electron like alkali metals to form
positive ion
c) as Hydrogen can gain one electron easily like Halogens to form
negative ion
d) as Hydrogen shows the properties of non-metals
18 (iii) Which of the following has highest electronegativity?
a) F
b) Cl
c) Br
d) I
18 (iv) Identify the reason for the gradual change in electronegativity in halogens down the group.
a) Electronegativity increases down the group due to decrease in atomic size
b) Electronegativity decreases down the group due to decrease in tendency to lose electrons
c) Electronegativity decreases down the group due to increase in atomic radius/ tendency to gain electron decreases
d) Electronegativity increases down the group due to increase in forces of attractions between nucleus & valence electrons
18 (v) Which of the following reason correctly justifies that “Fluorine (72pm) has smaller atomic radius than Lithium (152pm)”?
a) F and Li are in the same group. Atomic size increases down the group
b) F and Li are in the same period. Atomic size increases across the
period due to increase in number of shells
c) F and Li are in the same group. Atomic size decreases down the group
d) F and Li are in the same period and across the period atomic size/radius
decreases from left to right.
Answer:
Q18 (i)
Correct Option: (c) Cs > Rb > K > Na > Li
Reason:
Metallic character increases down the group because atomic size increases and atoms lose electrons more easily.
Q18 (ii)
Correct Option: (b) as Hydrogen can easily lose one electron like alkali metals to form positive ion
Reason:
Hydrogen has one electron and can lose it to form H⁺, similar to alkali metals.
Q18 (iii)
Correct Option: (a) F (Fluorine)
Reason:
Fluorine has the highest electronegativity due to its small atomic size and strong attraction for electrons.
Q18 (iv)
Correct Option: (c) Electronegativity decreases down the group due to increase in atomic radius / tendency to gain electron decreases
Reason:
As atomic size increases down the group, the attraction between nucleus and electrons decreases, so electronegativity decreases.
Q18 (v)
Correct Option: (d) F and Li are in the same period and across the period atomic size decreases from left to right
Reason:
Across a period, nuclear charge increases, pulling electrons closer, so atomic radius decreases from Li → F.
19. Read the following and answer any four questions from 19 (i) to 19 (v) Sumati wanted to see the stars of the night sky. She knows that she needs a telescope to see those distant stars. She finds out that the telescopes, which are made of lenses, are called refracting telescopes and the ones which are made of
mirrors are called reflecting telescopes.So she decided to make a refracting telescope. She bought two lenses, L1 and L2. out of which L1 was bigger and L2 was smaller. The larger lens gathers and bends the light, while the smaller lens magnifies the image. Big, thick lenses are more powerful. So to see far away, she needed a big powerful lens. Unfortunately, she realized that a big lens is very heavy. Heavy lenses are hard to make and difficult to hold in the right place. Also
since the light is passing through the lens, the surface of the lens has to be extremely smooth. Any flaws in the lens will change the image. It would be like looking through a dirty window.
19 (i) Based on the diagram shown, what kind of lenses would Sumati need to make
the telescope?
a) Concave lenses
b) Convex lenses
c) Bifocal lenses
19 (ii) If the powers of the lenses L1 and L2 are in the ratio of 4:1, what would be the
ratio of the focal length of L1 and L2?
a) 4:1
b) 1:4
c) 2:1
d) 1:1
19 (iii) What is the formula for magnification obtained with a lens?
a) Ratio of height of image to height of object
b) Double the focal length.
c) Inverse of the radius of curvature.
d) Inverse of the object distance.
19 (iv) Sumati did some preliminary experiment with the lenses and found out that the
magnification of the eyepiece (L2) is 3. If in her experiment with L2 she found
an image at 24 cm from the lens, at what distance did she put the object?
a) 72 cm
b) 12 cm
c) 8 cm
d) 6 cm
19 (v) Sumati bought not-so-thick lenses for the telescope and polished them. What
advantages, if any, would she have with her choice of lenses?
a) She will not have any advantage as even thicker lenses would give
clearer images.
b) Thicker lenses would have made the telescope easier to hand
c) Not-so-thick lenses would not make the telescope very heavy and also
allow considerable amount of light to pass.
d) Not-so-thick lenses will give her more magnification.
Answer:
Q19 (i)
Correct Option: (b) Convex lenses
Reason:
Refracting telescopes use convex lenses. The objective lens collects light and the eyepiece magnifies the image.
Q19 (ii)
Correct Option: (b) 1 : 4
Reason:
Power (P) = 1/f
So, focal length is inversely proportional to power.
If power ratio = 4 : 1, then focal length ratio = 1 : 4.
Q19 (iii)
Correct Option: (a) Ratio of height of image to height of object
Reason:
Magnification (m) = height of image / height of object.
Q19 (iv)
Correct Option: (c) 8 cm
Reason:
Magnification = v/u
Given m = 3 and v = 24 cm
So, 3 = 24 / u ⇒ u = 8 cm
Q19 (v)
Correct Option: (c)
Not-so-thick lenses would not make the telescope very heavy and also allow considerable amount of light to pass.
Reason:
Thin lenses are lighter, easier to handle, and still allow sufficient light, making them practical for telescopes.
20.Read the following and answer any 4 questions from 20 (i) to 20 (v).
A solenoid is a long helical coil of wire through which a current is run in order to create a magnetic field. The magnetic field of the solenoid is the superposition of the fields due to the current through each coil. It is nearly uniform inside the solenoid and close to zero outside and is similar to the field of a bar magnet having a north pole at one end and a south pole at the other depending upon the direction of current flow. The magnetic field produced in the solenoid is dependent on a few factors such as, the current in the coil, number of turns per unit length etc. The following graph is obtained by a researcher while doing an experiment to see the variation of the magnetic field with respect to the current in the solenoid.
The unit of magnetic field as given in the graph attached is in milli-Tesla (mT) and the current is given in Ampere.
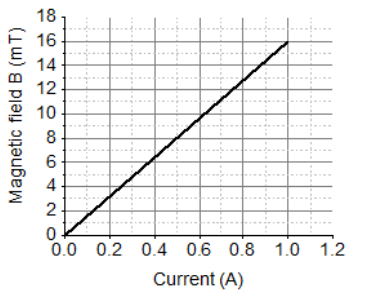
20. Read the following and answer any 4 questions from 20 (i) to 20 (v).
A solenoid is a long helical coil of wire through which a current is run in order to create a magnetic field. The magnetic field of the solenoid is the superposition of the fields due to the current through each coil. It is nearly uniform inside the solenoid and close to zero outside and is similar to the field of a bar magnet having a north pole at one end and a south pole at the other depending upon the direction of current flow. The magnetic field produced in the solenoid is dependent on a few factors such as, the current in the coil, number of turns per unit length etc.
The following graph is obtained by a researcher while doing an experiment to see the variation of the magnetic field with respect to the current in the solenoid.
The unit of magnetic field as given in the graph attached is in milli-Tesla (mT)
and the current is given in Ampere.
20 (i) What type of energy conversion is observed in a linear solenoid?
a. Mechanical to Magnetic
b. Electrical to Magnetic
c. Electrical to Mechanical
d. Magnetic to Mechanical
20 (ii) What will happen if a soft iron bar is placed inside the solenoid?
a. The bar will be electrocuted resulting in short-circuit.
b. The bar will be magnetised as long as there is current in the circuit.
c. The bar will be magnetised permanently.
d. The bar will not be affected by any means.
20 (iii) The magnetic field lines produced inside the solenoid are similar to that of …
a. a bar magnet
b. a straight current carrying conductor
c. a circular current carrying loop d. electromagnet of any shape
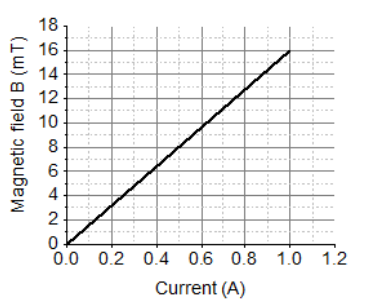
I. The magnetic field produced by the solenoid is inversely proportional to the current.
II. The magnetic field produced by the solenoid is directly proportional to the current.
III. The magnetic field produced by the solenoid is directly proportional to square of the current.
IV. The magnetic field produced by the solenoid is independent of the current.
Choose from the following which of the following would be the correct statement(s).
a. Only IV
b. I and III and IV
c. I and II
d. Only II
20 (v) From the graph deduce which of the following statements is correct.
a. For a current of 0.8A the magnetic field is 13 mT
b. For larger currents, the magnetic field increases non-linearly.
c. For a current of 0.8A the magnetic field is 1.3 mT
d. There is not enough information to find the magnetic field
corresponding to 0.8A current
Answer:
Q20 (i) What type of energy conversion is observed in a linear solenoid?
Correct Option: (b) Electrical to Magnetic
Reason: A solenoid converts electrical energy into magnetic energy by producing a magnetic field when current flows.
Q20 (ii) What will happen if a soft iron bar is placed inside the solenoid?
Correct Option: (b) The bar will be magnetised as long as there is current in the circuit.
Reason: Soft iron becomes a temporary magnet and loses magnetism when current stops.
Q20 (iii) The magnetic field lines produced inside the solenoid are similar to that of …
Correct Option: (a) a bar magnet
Reason: The magnetic field pattern of a solenoid is similar to that of a bar magnet.
Q20 (iv) After analysing the graph which statement is correct?
Correct Option: (d) Only II
Reason: Magnetic field is directly proportional to current (B ∝ I).
Q20 (v) From the graph deduce the correct statement.
Correct Option: (c) For a current of 0.8 A the magnetic field is 1.3 mT
Reason: The graph shows a linear relation, and at 0.8 A the value is 1.3 mT.
SECTION B
21 Bile juice does not have any digestive enzyme but still plays a significant role in the process of digestion. Justify the statement.
OR
In birds and mammals the left and right side of the heart are separated. Give reasons.
Answer::
Bile juice helps in digestion by emulsifying fats and making the medium alkaline.
Reason:
It breaks large fat globules into smaller droplets, increasing surface area for enzyme action and also neutralises the acidic food coming from the stomach.
22 State the events occurring during the process of photosynthesis. Is it essential that these steps take place one after the other immediately?
Answer:
The process of photosynthesis consists of three main events:
- Reduction of carbon dioxide to carbohydrates (like glucose).
- Absorption of light energy by chlorophyll.
- Conversion of light energy to chemical energy and splitting of water molecules into hydrogen and oxygen.
23 Give a test that can be used to confirm the presence of carbon in a compound. With a valency of 4, how is carbon able to attain noble gas configuration in its compounds?
OR
The number of carbon compounds is more than those formed by all other elements put together. Justify the statement by giving two reasons.
Answer:
Test for Carbon
Burn the compound in air. If it produces a gas that turns lime water milky, it confirms the presence of carbon (due to the formation of CO2).
Attaining Noble Gas Configuration
Carbon has 4 valence electrons. It cannot gain or lose 4 electrons because of the high energy requirements. Instead, it shares its 4 valence electrons with other atoms to form covalent bonds, thereby completing its octet.
Justification for Large Number of Compounds
Carbon forms more compounds than all other elements combined due to:
- Catenation: The unique ability of carbon to form stable, long chains or rings by bonding with other carbon atoms.
- Tetravalency: Having a valency of 4 allows carbon to bond with a wide variety of other elements (like H, O, N, and Cl) in various orientations.
24 The following observations were made by a student on treating four metals P, Q, R and S with the given salt solutions:
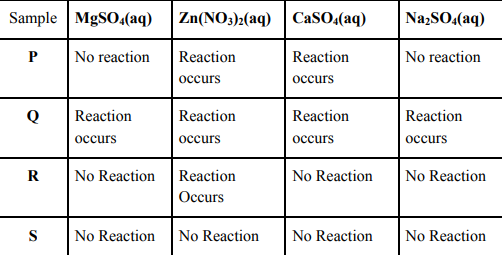
Based on the above observations:
(a) Arrange the given samples in the increasing order of reactivity
(b) Write the chemical formulae of products formed when Q reacts with CuSO4 solution.
25
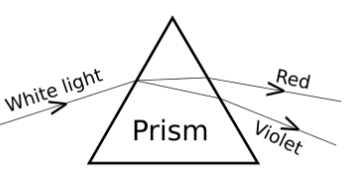
A student observes the above phenomenon in the lab as a white light passes through a prism. Among many other colours, he observed the position of the two colours Red and Violet.
What is the phenomenon called? What is the reason for the violet light to bend
more than the red light?
26.
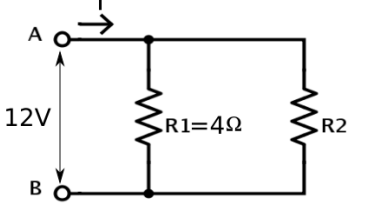
A student has two resistors- 2 Ω and 3 Ω. She has to put one of them in place of R2 as shown in the circuit. The current that she needs in the entire circuit is exactly 9A. Show by calculation which of the two resistors she should choose.
Section C
27 After self-pollination in pea plants with round, yellow seeds, following types of seeds were obtained by Mendel:
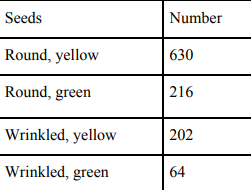
Analyze the result and describe the mechanism of inheritance which explains these results.
OR
In humans, there is a 50% probability of the birth of a boy and 50 % probability that a girl will be born. Justify the statement on the basis of the mechanism of sex-determination in human beings.
28 Plastic cups were used to serve tea in trains in early days- these could be returned to the vendors, cleaned and reused. Later,Kulhads were used instead of plastic cups. Now, paper cups are used for serving tea. What are the reasons for the shift from Plastic to Kulhads and then finally to paper cups?
29 Explain where and how urine is produced? 3
30 a. Which of the following reactions is/ are an endothermic reaction(s) where decomposition also happens?
- Respiration
- Heating of lead nitrate
- Decomposition of organic matter
- Electrolysis of acidified water
b. Silver chloride when kept in the open turns grey. Illustrate this with a balanced chemical equation.
31 The following table shows the position of five elements A, B, C, D and E in
the modern periodic table.
Answer the following giving reasons:
(i) Which element is a metal with valency two?
(ii) Which element is least reactive?
(iii) Out of D and E which element has a smaller atomic radius?
32 a. Explain the formation of Calcium Chloride with the help of electron dot structure. (At numbers: Ca = 20; Cl = 17)
b. Why do ionic compounds not conduct electricity in solid state but conduct electricity in molten and aqueous state?
32 (a) Formation of Calcium Chloride (CaCl₂) using electron dot structure
- Atomic number of Ca = 20 → electronic configuration = 2, 8, 8, 2 → 2 valence electrons
- Atomic number of Cl = 17 → electronic configuration = 2, 8, 7 → 7 valence electrons
Process:
- Calcium loses its 2 valence electrons:
- Each chlorine atom gains 1 electron:
So, one Ca atom transfers one electron to each of two Cl atoms → forming Ca²⁺ and 2Cl⁻ ions.
Electron dot representation (conceptually):
- Ca has 2 dots → transfers both
- Each Cl has 7 dots → gains 1 to complete octet
Final structure:
32 (b) Electrical conductivity of ionic compounds
- In solid state:
- Ions are tightly packed in a fixed lattice structure.
- They cannot move freely, so no conduction of electricity.
- In molten or aqueous state:
- Ionic compounds dissociate into free-moving ions.
- These mobile ions carry electric charge.
- Hence, they conduct electricity.
33. Refractive index of water with respect to air is 1.33 and that of diamond is 2.42.
(i) In which medium does the light move faster, water or diamond?
(ii) What is the refractive index of diamond with respect to water? 3
(i) The speed of light in a medium is inversely proportional to its refractive index ( v=nc ).
Since water has refractive index 1.33 and diamond has 2.42, light travels faster in water (lower refractive index → higher speed).
(ii) Refractive index of diamond with respect to water is:ndiamond w.r.t water=nwaterndiamond=1.332.42≈1.82
Section D
34 Match the following pH values 1, 7, 10, 13 to the solutions given below:
- Milk of magnesia
- Gastric juices
- Brine
- Aqueous Sodium hydroxide.
Amit and Rita decided to bake a cake and added baking soda to the cake batter.
Explain with a balanced reaction, the role of the baking soda. Mention any other use of baking soda.
OR
(i) Four samples A, B, C and D change the colour of pH paper or solution to Green, Reddish-pink, Blue and Orange. Their pH was recorded as 7, 2, 10.5 & 6 respectively. Which of the samples has the highest amount of Hydrogen ion concentration? Arrange the four samples in the decreasing order of their pH.
(ii) Rahul found that the Plaster of Paris, which he stored in a container, has become very hard and lost its binding nature. What is the reason for this? so, write a chemical equation to represent the reaction taking place.
(iii) Give any one use of Plaster of Paris other than for plastering or smoothening of walls.
Ans: When Amit and Rita added baking soda (sodium hydrogen carbonate, NaHCO₃) to the cake batter, the following happens:
- On heating, baking soda decomposes:
- The carbon dioxide (CO₂) gas produced forms bubbles in the batter.
- These bubbles get trapped, causing the cake to rise and become soft and fluffy.
- The water vapor also helps in making the cake light.
Conclusion: Baking soda helps the cake become spongy and fluffy due to the release of carbon dioxide gas during heating.
35. Trace the changes that take place in a flower from gamete formation to fruit formation. 5
Ans: The transformation of a flower into a fruit involves several stages:
1. Gamete Formation
- In the anther, male gametes are formed inside pollen grains.
- In the ovary, the ovule forms the female gamete (egg cell).
2. Pollination
- Transfer of pollen grains from anther to stigma (by wind, insects, etc.).
3. Pollen Germination
- Pollen grain germinates on the stigma and forms a pollen tube.
- The tube grows through the style toward the ovule.
4. Fertilization
- Male gamete travels through the pollen tube and fuses with the egg → forms zygote.
- In flowering plants, double fertilization occurs:
- One male gamete + egg → zygote
- Other male gamete + polar nuclei → endosperm
5. Formation of Seed and Fruit
Other floral parts (petals, sepals, stamens) usually wither away.
Zygote develops into an embryo.
Ovule becomes the seed.
Ovary develops into the fruit.
36 In the given circuit, A, B, C and D are four lamps connected with a battery of 60V
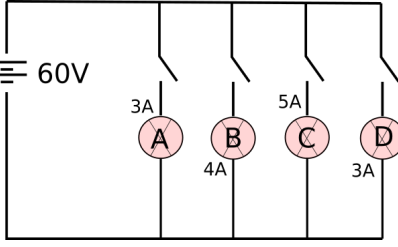
Analyse the circuit to answer the following questions.
(i) What kind of combination are the lamps arranged in (series or parallel)?
(ii) Explain with reference to your above answer, what are the advantages (any
two) of this combination of lamps?
(iii) Explain with proper calculations which lamp glows the brightest?
(iv) Find out the total resistance of the circuit.
OR
PQ is a current carrying conductor in the plane of the paper as shown in the
figure below.
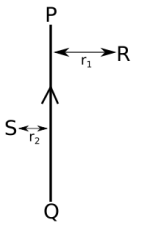
(i) Find the directions of the magnetic fields produced by it at points R and S?
(ii) Given r1> r2, where will the strength of the magnetic field be larger? Give reasons.
(iii) If the polarity of the battery connected to the wire is reversed, how would the direction of the magnetic field be changed?
(iv) Explain the rule that is used to find the direction of the magnetic field for a straight current carrying conductor.
SECTION B – CBSE Class 10 Science Sample Paper 2020–21

📘 Math & Science Solutions by Class
🔹 Class 10
🔹 Class 9
🔹 Class 8
🔹 Class 7
🔹 Class 6
🔹 Class 12
🔹 Class 11
- Class 11 Math Solutions
- Class 11 Physics Solutions
- Class 11 Chemistry Solutions
- Class 11 Biology Solutions
For the official Class 10 Mathematics Solutions, you can visit:
- NCERT Textbooks (for Class 10):
The CBSE Class 10 Science Sample Paper 2020–21 plays a vital role in helping students practice and revise key concepts from Physics, Chemistry, and Biology. With a variety of question types—from one-mark MCQs to five-mark long answers—this paper helps students prepare strategically for the board exam. By working through each solved question, learners can assess their understanding and gain clarity on how to present answers as expected by CBSE.
Solving the CBSE Class 10 Science Sample Paper 2020–21 not only boosts confidence but also improves time management during the actual exam. Since the paper follows the updated format introduced during the pandemic period, it is an ideal resource for understanding the structure of board papers in a new pattern.
Every question in the CBSE Class 10 Science Sample Paper 2020–21 is aligned with NCERT concepts and the official syllabus, making it a reliable tool for revision. Whether it’s numerical problems in electricity, explanation-based questions in biology, or chemical reactions in chemistry, this paper covers all essential areas of the Class 10 Science curriculum.
In summary, the CBSE Class 10 Science Sample Paper 2020–21 offers complete exam readiness. Students are advised to solve it thoroughly, compare with detailed solutions, and learn the correct way to attempt answers for maximum marks. Regular practice of such sample papers ensures better performance and reduces exam-day stress.
