Grade 11 Physics Thermodynamics NCERT Solution helping students understand how heat, work, and internal energy are related in physical systems. This chapter introduces fundamental concepts such as the zeroth law, first law of thermodynamics, heat capacity, specific heat, adiabatic and isothermal processes, and heat engines.
In this page, you will find easy-to-understand NCERT solutions for all questions of the Thermodynamics chapter. Each numerical is solved step by step, with clear explanations and properly applied formulas. Conceptual questions are also answered in simple language so that every student can understand them without difficulty.
These solutions will not only help you in CBSE exams but also build a strong foundation for competitive exams like NEET, JEE Main, and JEE Advanced.
Grade 11 Physics Thermodynamics NCERT Solution
Question 11.1
A geyser heats water flowing at the rate of 3.0 litres per minute from 27 degrees C to 77 degrees C. If the geyser operates on a gas burner whose heat of combustion is 4.0 × 10^4 J/g, find the rate of consumption of the fuel.
Solution :
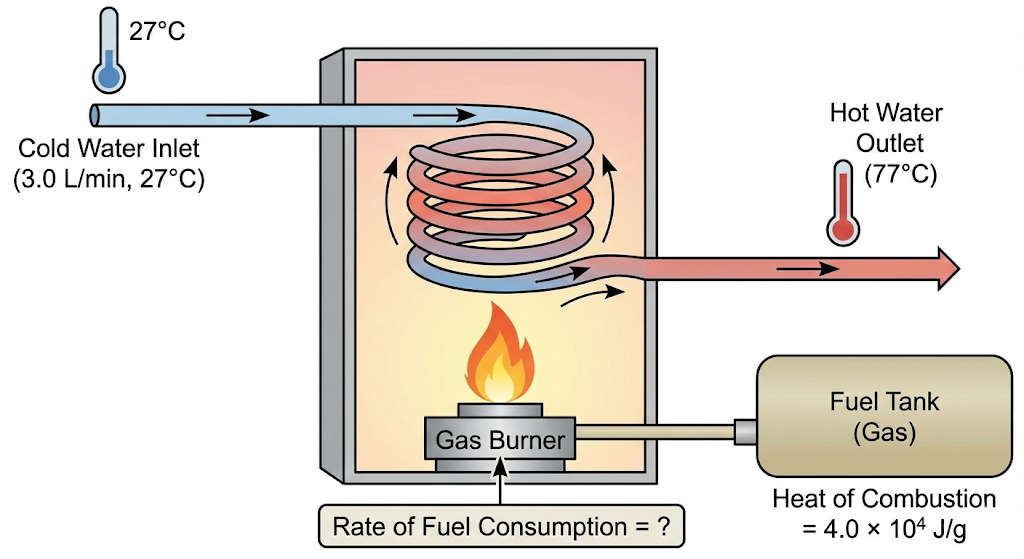
Given:
Flow rate of water = 3.0 litres/min
Since density of water ≈ 1 kg/L,
Mass of water per minute
m = 3.0 kg/min
Initial temperature, T₁ = 27°C
Final temperature, T₂ = 77°C
Temperature rise,
ΔT = 77 − 27 = 50°C
Specific heat capacity of water,
c = 4.2 × 10³ J/kg°C
Heat of combustion of fuel = 4.0 × 10⁴ J/g
Step 1: Heat required per minute
Formula:
Q = mcΔT
Substituting:
Q = 3.0 × (4.2 × 10³) × 50
Q = 3 × 4200 × 50
Q = 630000 J
Q = 6.3 × 10⁵ J per minute
Step 2: Fuel required per minute
Heat released by 1 gram fuel = 4.0 × 10⁴ J
Fuel consumption rate:
Question 2
What amount of heat must be supplied to 2.0 × 10^-2 kg of nitrogen (at room temperature) to raise its temperature by 45 degrees C at constant pressure? (Molecular mass of N2 = 28; R = 8.3 J/mol·K)
Solution :
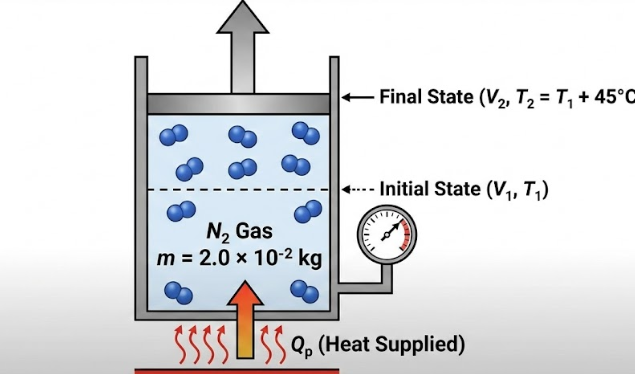
Mass of nitrogen,
m = 2.0 × 10⁻² kg Molar mass of N₂ = 28 g/mol = 28 × 10⁻³ kg/mol
Temperature rise,
ΔT = 45°C = 45 K
Gas constant,
R = 8.3 J/mol·K
Process: Constant pressure
Step 1: Find number of moles
Step 2: Molar specific heat at constant pressure
Nitrogen (N₂) is a diatomic gas at room temperature.
For a diatomic gas:
Step 3: Heat supplied
Formula:
Question 3
Explain why:
(a) Two bodies at different temperatures T1 and T2, if brought into thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
(b) The coolant in a chemical or nuclear plant should have high specific heat.
(c) Air pressure in a car tyre increases during driving.
(d) The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
Solution (explanations with examples)
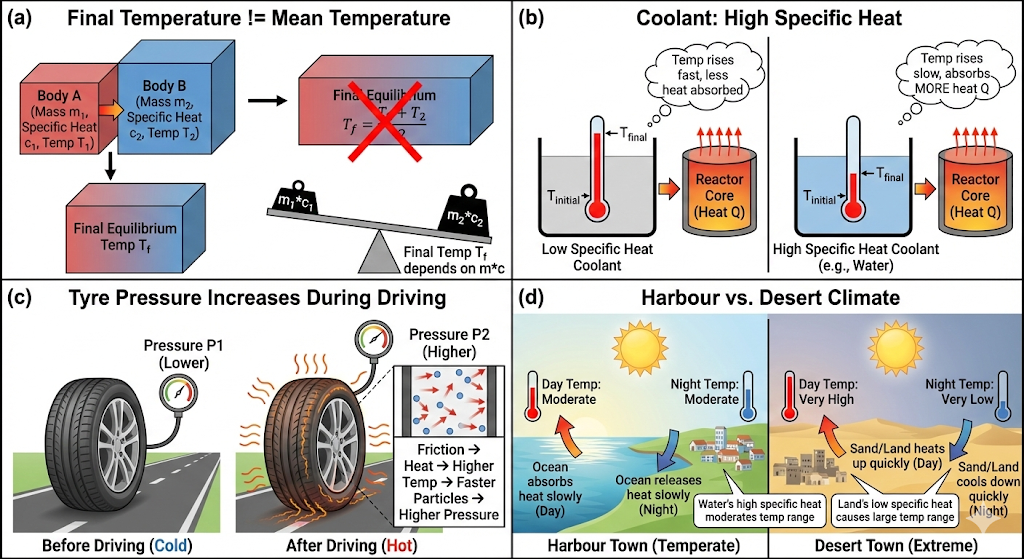
(a) Two bodies at different temperatures T₁ and T₂ do not necessarily settle at
When two bodies are brought into thermal contact, the final temperature depends on their masses and specific heat capacities, not just their initial temperatures.
Heat lost by hot body = Heat gained by cold body
If the two bodies have equal mass and equal specific heat, only then:
Otherwise, the final temperature will be closer to the body having larger heat capacity (mc).
(b) The coolant in a chemical or nuclear plant should have high specific heat
Specific heat is the amount of heat required to raise the temperature of 1 kg of a substance by 1°C.
A coolant with high specific heat:
- Absorbs a large amount of heat
- Shows only a small rise in temperature
- Prevents overheating
For example, water is commonly used because it has high specific heat capacity.
Thus, it removes large heat safely and efficiently.
(c) Air pressure in a car tyre increases during driving
During driving:
- Tyres rub against the road
- Air inside the tyre gets heated
As temperature increases, according to the gas law:
So, when temperature rises, pressure increases.
Therefore, tyre pressure becomes higher during driving.
(d) Climate of a harbour town is more temperate than that of a desert town at the same latitude
Water has high specific heat capacity, while sand has low specific heat capacity.
In harbour towns:
- Sea water heats slowly during the day
- Sea water cools slowly at night
This reduces temperature variation.
In desert areas:
- Sand heats quickly during the day
- Sand cools quickly at night
This causes large temperature variation.
Hence, harbour towns have moderate (temperate) climate, while desert towns have extreme climate.
Question 11.4
A cylinder with a movable piston contains hydrogen at standard temperature and pressure. The walls of the cylinder are insulating and the piston is insulated by a pile of sand. By what factor does the pressure of the gas increase if the gas is compressed to half its original volume? (Assume adiabatic compression of diatomic hydrogen.)
Solution :
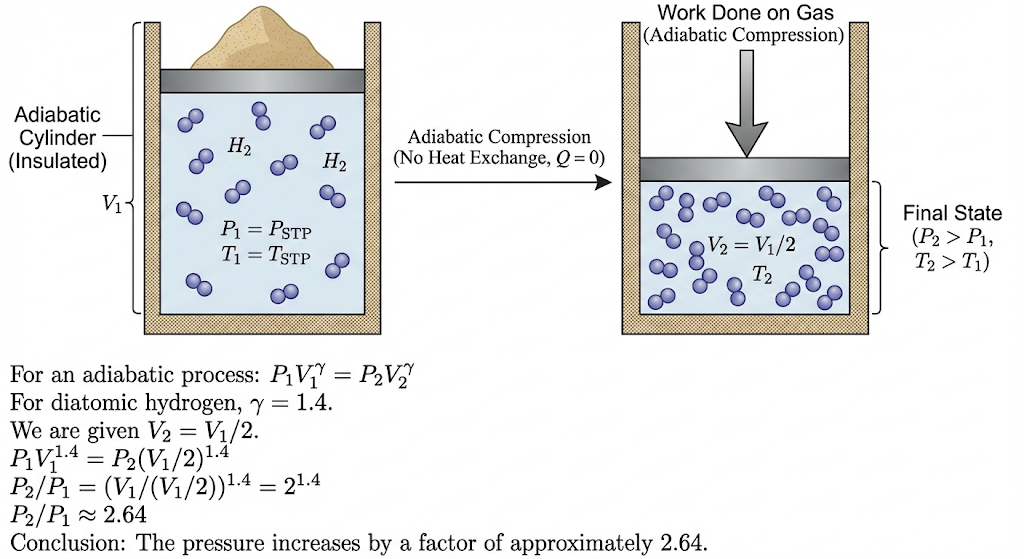
This is an adiabatic compression problem.
Since the walls and piston are insulated → no heat exchange → process is adiabatic.
For an adiabatic process:
Step 1: Value of γ for hydrogen
Hydrogen (H₂) is a diatomic gas.
For a diatomic gas:
Step 2: Use adiabatic relation
Given:
So,
Step 3: Substitute γ = 1.4
Final Answer:
So, the pressure increases by a factor of about 2.6.
Question 11.5
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J.)
Solution :
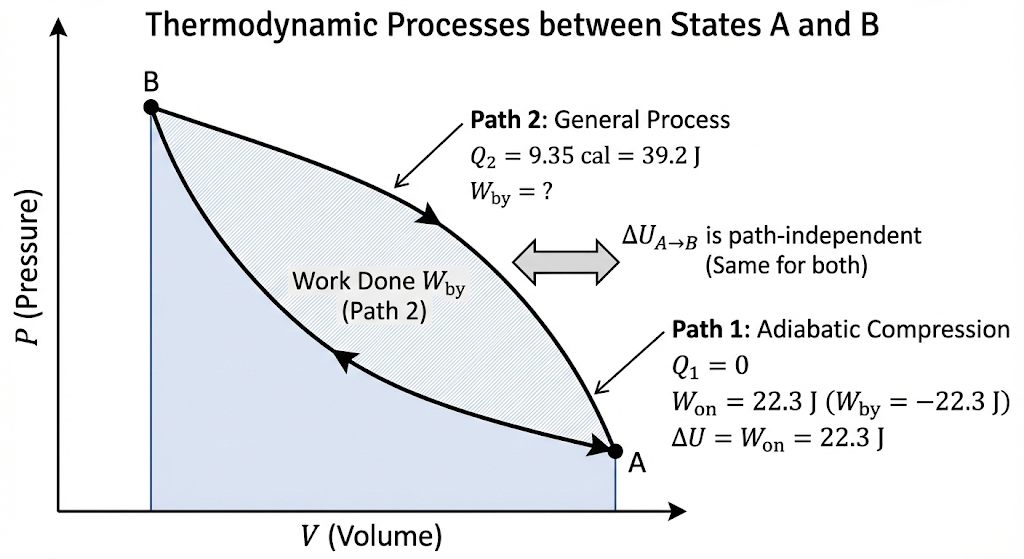
This is a direct application of the First Law of Thermodynamics.
Step 1: Find change in internal energy (ΔU)
In the first process (adiabatic):
Adiabatic ⇒
Given:
Work done on the system = 22.3 J
Work done by the system = −22.3 J
Using first law:
So,
Step 2: Second process (A → B via different path)
Heat absorbed:
Convert into joules:
Step 3: Apply First Law again
Final Answer:
So, the net work done by the system in the second process is about 17 J.
Question 11.6
Two cylinders A and B of equal capacity are connected to each other via a stopcock. Cylinder A contains a gas at standard temperature and pressure. Cylinder B is completely evacuated (vacuum). The entire system is thermally insulated. The stopcock is suddenly opened. Answer:
(a) What is the final pressure of the gas in A and B?
(b) What is the change in internal energy of the gas?
(c) What is the change in the temperature of the gas?
(d) Do the intermediate states of the system (before settling to the final equilibrium) lie on its P-V-T surface?
Solution :
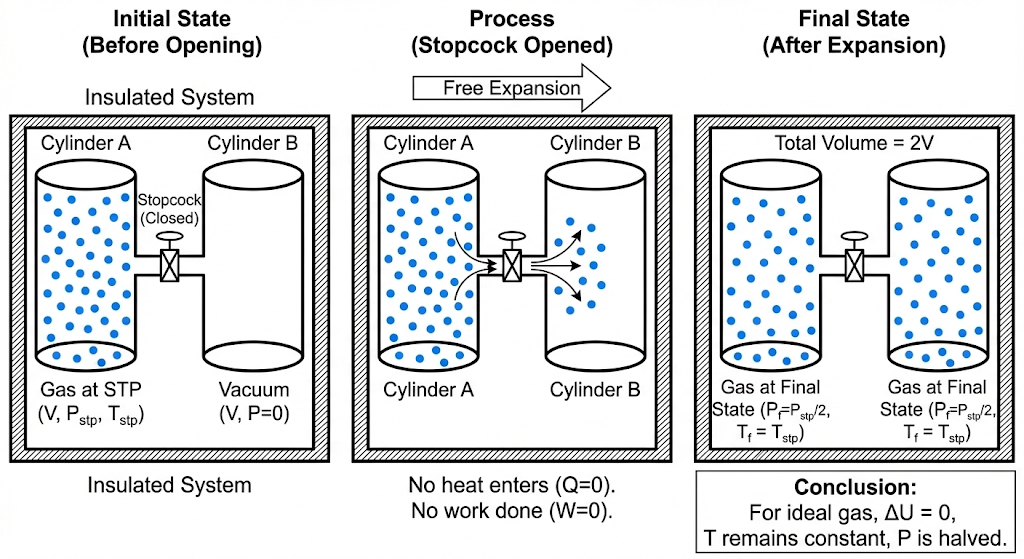
Let us solve it carefully using the First Law of Thermodynamics:
Step 1: First process (Adiabatic)
For an adiabatic process:
Given:
work done by the system:
Now,
So the change in internal energy between states A and B is:
Step 2: Second process (Different path)
Heat absorbed:
Convert into joules:
Step 3: Apply First Law again
Question 11.7
An electric heater supplies heat to a system at a rate of 100 W. If the system performs work at a rate of 75 J per second, at what rate is the internal energy increasing?
Solution :
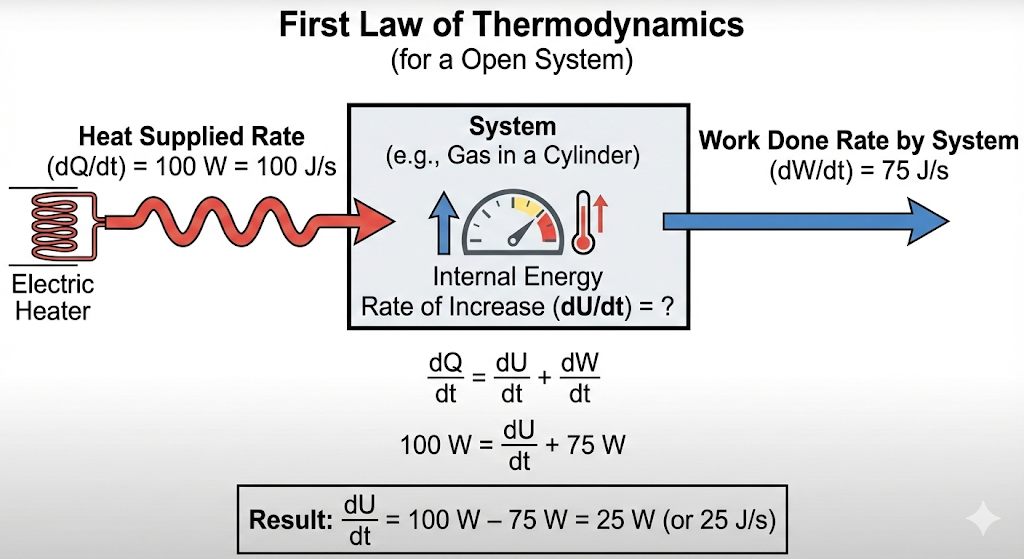
Use the First Law of Thermodynamics:
Given:
Heat supplied per second:
Work done by system per second:
Apply First Law (rate form):
Final Answer:
Rate of increase of internal energy=25 J/s
So, the internal energy is increasing at a rate of 25 W (or 25 J per second).
11.8
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in the figure. The important points on the P–V plot are Its volume is then reduced from E to F by an isobaric process (pressure = 300 N/m²). Calculate the total work done by the gas for the process D → E → F.
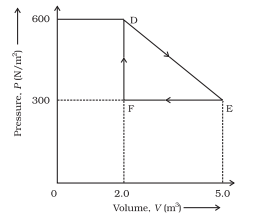
From the P–V diagram, the important points are:
- D: V=2m3, P=600N/m2
- E: V=5m3, P=300N/m2
- F: V=2m3, P=300N/m2
We must calculate total work done in the process:
Step 1: Work done from D → E
This is a linear process, so work equals area under the straight line in the P–V graph.
Area of trapezium:
So,
(Positive because volume increases — expansion)
Step 2: Work done from E → F
This is an isobaric compression at
Volume decreases from 5 m³ to 2 m³.
(Negative because compression — work done on gas)
Step 3: Total Work
For the official Class 11 Physics Solutions, you can visit:
- NCERT Textbooks
Class-wise Solutions
Class 12:
Class 12 Physics – NCERT Solutions
Class 12 Chemistry – NCERT Solutions
Class 11:
- Class 11 Physics – NCERT Solutions
- Class 11 Chemistry – NCERT Solutions
- Class 11 Biology – NCERT Solutions
- Class 11 Math – NCERT Solutions
Class 10:
Class 9:
Class 8:
Class 7:
Class 6:
Subject-wise Solutions
Physics:
Chemistry:
Biology:
Math:
- Class 11 Math – NCERT Solutions
- Class 10 Math – NCERT Solutions
- Class 9 Math – NCERT Solutions
- Class 8 Math – NCERT Solutions
Science:
- Class 10 Science – NCERT Solutions
- Class 9 Science – NCERT Solutions
- Class 8 Science – Oxford Solutions
- Class 7 Science – Oxford Solutions
- Class 6 Science – Oxford Solutions
